A trial looking at a vaccine, chemotherapy and an immunotherapy for cancer that has spread from where it started (MAGE Vaccine Trial)
Please note - this trial is no longer recruiting patients. We hope to add results when they are available.
Cancer type:
Status:
This trial is looking at 3 vaccines with standard treatment for non small cell lung cancer, oesophageal cancer and gastro oesophageal junction cancer that has spread from where it started.
Cancer Research UK supports this trial.
More about this trial
There are  for advanced:
for advanced:
- non small cell lung cancer (NSCLC)
- cancer of the food pipe (oesophageal cancer)
- cancer where the food pipe meets the stomach (gastro oesophageal junction cancer)
One standard treatment for these cancers is chemotherapy and an  called pembrolizumab.
called pembrolizumab.
In this trial researchers are looking at the ChAdOx1-MAGEA3-NYESO vaccine, the MVA-MAGE-A3 and the MVA-NYESO vaccines.
MAGE-A3 and NYESO-1 are proteins found on some cancer cells. Not everyone has them and not every cancer type has these proteins.
The vaccines contain harmless parts of the MAGE-A3 and NYESO-1 proteins. The vaccines work by helping the  find the proteins. The immune system recognizes them as something foreign in the body and attacks them.
find the proteins. The immune system recognizes them as something foreign in the body and attacks them.
 have shown that the vaccines help the immune system recognize and attack cancer cells with these proteins.
have shown that the vaccines help the immune system recognize and attack cancer cells with these proteins.
Researchers want to find out if this happens when people with cancer have the vaccines.
There are 2 parts to this trial. In the first part a small number of people have the vaccines with the standard treatment. The team want to find out how safe it is to give them with standard treatment.
In the second part people with NSCLC have either the:
- standard treatment by itself
- standard treatment and the vaccines
In the second part people with oesophageal cancer or gastro oesophageal junction cancer have the standard treatment with the vaccines.
The aims of this trial are to find out:
- how safe it is to have the vaccines with standard treatment
- how acceptable it is
- how well the vaccines work with standard treatment
- what happens in the body after having the vaccines
- more about the side effects
Who can enter
The following bullet points are a summary of the entry conditions for this trial. Talk to your doctor or the trial team if you are unsure about any of these. They will be able to advise you.
Who can take part
You may be able to join this trial if you have:
squamous cell  non small cell lung cancer (NSCLC) that is over 5 cm across, has spread to lymph nodes or nearby organs (stage 3B or 3C). Or your cancer has spread to both lungs or another part of the body (stage 4). You can also join if you have a non squamous NSCLC that is stage 3B or stage 4.
non small cell lung cancer (NSCLC) that is over 5 cm across, has spread to lymph nodes or nearby organs (stage 3B or 3C). Or your cancer has spread to both lungs or another part of the body (stage 4). You can also join if you have a non squamous NSCLC that is stage 3B or stage 4.
Or
- squamous cell cancer of the food pipe (oesophagus) or where the food pipe joins the stomach (gastro oesophageal junction) that cannot be removed by surgery and has spread into nearby tissue (stage 3) or has spread to another part of the body (stage 4)
Also all of the following must apply. You:
- are to have pembrolizumab and chemotherapy but you have not started treatment yet
- have an area of cancer that the doctor can measure
- have a tissue sample
- have cancer cells with the protein PD-L1 and the protein (
biomarker  ) called MAGE-A3
) called MAGE-A3 - are fit and active but might not be able to do heavy physical work (performance status 0 or 1)
- have satisfactory blood test results
- are willing to use 2 types of contraception during treatment and for a time after if you or your partner could become pregnant
- are at least 18 years old
Who can’t take part
Cancer related
You cannot join this trial if any of these apply. You:
- have had chemotherapy and or radiotherapy for your NSCLC unless it was completed 6 months before your diagnosis of cancer spread
- have already had an
immunotherapy 
- have or had another cancer apart from successfully treated
non melanoma skin cancer  ,
, early cancer  (non muscle invasive) of the bladder or a
(non muscle invasive) of the bladder or a carcinoma in situ 
- have cancer spread to the brain, spinal cord or tissue surrounding the brain. You might be able to join if it is treated, has not got worse and doesn’t need treatment.
Medical conditions
You cannot join this trial if any of these apply. You:
- have had major chest surgery or tummy (abdominal) surgery and have not recovered fully
- have a heart trace (
ECG  ) that shows your heart rhythm isn’t normal. Your doctor will test for this and tell you the results.
) that shows your heart rhythm isn’t normal. Your doctor will test for this and tell you the results. - have an active infection or similar disease that affects your whole body
- have HIV, hepatitis B or hepatitis C
- have an
auto immune disease  apart from certain ones. Your doctor will know which ones these are.
apart from certain ones. Your doctor will know which ones these are. - have had the AstraZeneca COVID-19 vaccine (also called Vaxzevria) within 6 weeks of starting treatment
- have an
immune system  that isn’t working well or you are taking medication that damps down the immune system. This is apart from a small daily dose of steroids.
that isn’t working well or you are taking medication that damps down the immune system. This is apart from a small daily dose of steroids. - have had a stem cell transplant from a donor (allogeneic transplant) within the past 5 years. You might be able to join if your transplant was more than 5 years ago and you have no symptoms of
graft versus host disease  (GvHD)
(GvHD) - have had a medical condition that causes the fluid to leak from the tiny blood vessels (
capillary  leak syndrome)
leak syndrome) - have a history of having a low count of a blood cells called
platelets  after having
after having heparin  (This is HITT or HIT type 2.)
(This is HITT or HIT type 2.) - are taking part or going to take part in another clinical trial that uses an experimental drug
- have any other medical condition or mental health problem that could affect you taking part
Other
You cannot join this trial if any of these apply. You:
- are allergic to an immunotherapy, any ChAdOx1 or MVA vaccines or any of their ingredients
- have a severe allergy to eggs or to another vaccine
- have a
live vaccine  within in 4 weeks of joining the trial. The COVID-19 vaccines aren’t live vaccines. But you can’t have these vaccines within a certain time before and after the trial vaccine. Your doctor will know how long this is.
within in 4 weeks of joining the trial. The COVID-19 vaccines aren’t live vaccines. But you can’t have these vaccines within a certain time before and after the trial vaccine. Your doctor will know how long this is.
Trial design
This is a phase 1/2 trial. There are 2 parts to this trial.
Part 1
For this part of the trial the team needed 6 people to join. So far 5 people have joined.
The team want to find out that it is safe to have the vaccines with the standard treatment. To find this out the people are monitored for any side effects.
The team have found that they can give the MAGE-A3 vaccines with chemotherapy and pembrolizumab.
They are still looking at whether it is safe to give the MAGE-A3 and NY-ESO-1 vaccines with chemotherapy and pembrolizumab.
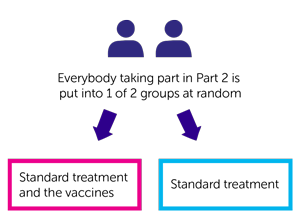
Part 2
This part of the trial is currently open to new patients. People in part 2 have the vaccines with the standard treatment. It is for people with:
- non small cell lung cancer
- oesophageal cancer
- gastro oesophageal junction cancer
People with non small cell lung cancer
The team need 80 with NSCLC people for part 2. This part is a randomised trial. A computer puts you into 1 of 2 groups. Neither you nor your doctor can choose which group you are in. The groups are:
- standard treatment and the vaccines
- standard treatment
Standard treatment
This is either:
- pembrolizumab, pemetrexed and cisplatin or carboplatin
- or pembrolizumab, paclitaxel and carboplatin
Your doctor will talk to about which is best treatment for you.
Pembrolizumab, pemetrexed and cisplatin or carboplatin
You have pembrolizumab, pemetrexed and cisplatin or carboplatin as a drip into a vein. You have them every 3 weeks. This is a  .
.
After 4 cycles of treatment you then have either:
- pembrolizumab every 3 weeks for about 2 years (35 cycles) or
- pembrolizumab and pemetrexed every 3 weeks for about 2 years (35 cycles) or
- a higher dose of pembrolizumab every 5 weeks for 2 years
Pembrolizumab, paclitaxel and carboplatin
You have pembrolizumab, paclitaxel and carboplatin as a drip into a vein. You have them on the same day every 3 weeks. This is a cycle of treatment. You have 4 cycles of treatment.
You then have pembrolizumab:
- at the same dose every 3 weeks for about 2 years (35 cycles) or
- at a higher dose every 6 weeks for up to 2 years
People with oesophageal or gastro oesophageal junction cancer
The team need up to 17 people with these cancers for part 2.
You have chemotherapy and pembrolizumab.
The chemotherapy you have can include:
You have oxaliplatin, cisplatin and 5FU as a drip into a vein. You have capecitabine as a tablet.
Your doctor will discuss with you what chemotherapy you have and how many cycles of treatment you have.
You have pembrolizumab as a drip into a vein every 3 weeks or 6 weeks with your chemotherapy. After finishing chemotherapy you then continue to have pembrolizumab:
- at the same dose every 3 weeks for about 2 years (35 cycles) or
- at a higher dose every 6 weeks for up to 2 years
You have the vaccines as an injection into a muscle. You have them in the muscle at the top of the arm, top of the leg or the buttocks. You have them in the same muscle all the time unless it becomes sore or there is a reaction.
You have a diary cards to complete every day. The team explains how to use them. You record details about:
- the injection site
- your general wellbeing
You must bring the cards with you when you attend your hospital appointments.
Samples for research
The team take extra blood samples during the trial. Where possible they take these when you have your routine bloods done.
Researchers use these samples to find out:
- how well the vaccines are working
- why the treatment might work better for some people
Hospital visits
You see the doctor to have tests before taking part. These tests include:
- a
physical examination 
- blood tests
- CT scan or an MRI scan
- urine test
- heart trace (ECG)
Everyone sees the doctor at regular times during treatment. This is for blood tests and to see how you are. You also have another CT scan or MRI scan.
Around your 10th cycle of treatment you see the doctor. This is for blood tests and to see how you are. This is your last visit for the trial.
If you have side effects caused by the vaccines you see the doctor every month until the side effects are gone. Where possible these will be at the same time you have your routine appointments.
Side effects
The trial team monitor you during treatment and afterwards. Contact your advice line or tell your doctor or nurse if any side effects are bad or not getting better.
This is the first time the ChAdOx1-MAGEA3-NYESO vaccine, MVA-MAGEA3 vaccine and MVA-NYESO vaccine are being used in humans. There might be side effects we don’t know about yet. Based on similar vaccines and laboratory studies the possible side effects might include:
- a reaction at the injection site such as redness, itching, swelling, feeling warm and a rash
- tiredness and lack of energy (fatigue)
- flu-like symptoms
- joint and muscle pain
- a drop in blood cells causing an increased risk of infection, bruising, bleeding and shortness of breath
- an allergic reaction
- changes to how well your heart works
- feeling or being sick
- diarrhoea
- tummy (abdominal) pain
- headaches
- dizziness
- feeling sleepy
- lack of energy
- loss of appetite
- swollen
lymph nodes 
|
Pembrolizumab can affect the These side effects could happen during treatment or months after treatment has finished. Rarely, these side effects could be life threatening. Your doctor or nurse can explain what these side effects are, the risk of them happening and what to look out for. If you have any of these side effects tell your doctor or nurse as soon as possible. You should tell them that you are on or have been on an immunotherapy. |
We have information about:
- pembrolizumab
- paclitaxel and carboplatin
- pemetrexed and cisplatin
- pemetrexed and carboplatin
- oxaliplatin
- capecitabine
- fluorouracil (5FU)
- cisplatin
Your doctor or a member of the trial team will talk to you about the possible side effects of the treatments before you agree to take part in the trial.
Recruitment start:
Recruitment end:
How to join a clinical trial
Please note: In order to join a trial you will need to discuss it with your doctor, unless otherwise specified.
Chief Investigator
Professor Fiona Blackhall
Supported by
Cancer Research UK
Other information
This is Cancer Research UK trial number CRUKD/20/001.
If you have questions about the trial please contact our cancer information nurses
Freephone 0808 800 4040